Ianalumab for immune thrombocytopenia
What is ianalumab for immune thrombocytopenia?
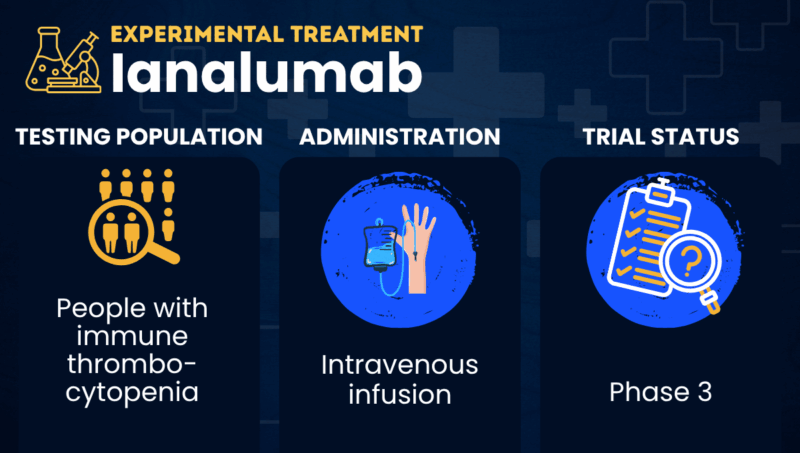
Ianalumab (VAY736) is an antibody-based therapy being explored as a potential treatment for immune thrombocytopenia (ITP).
In ITP, immune B-cells produce self-reactive antibodies that target platelets for destruction. Platelets are the small cell fragments that help form blood clots. When platelet levels are low, blood cannot clot properly, putting patients at an increased risk of bleeding.
Administered via an intravenous (into-the-vein) infusion, ianalumab is an antibody-based therapy designed to simultaneously lower the number and reduce the activity of B-cells. The therapy specifically achieves this by activating antibody-dependent cellular toxicity — an immune mechanism whereby cells covered in antibodies are flagged to be killed by certain immune cells — and by blocking the BAFF receptor, preventing B-cells from receiving signals that are crucial for their function and survival. Through this mechanism, ianalumab is expected to lower the levels of the harmful antibodies driving ITP, helping to control disease symptoms.
Novartis is exploring ianalumab’s therapeutic potential as a first- and second-line therapy for primary ITP, a type of ITP with no clear underlying causes. The therapy is currently being tested in a series of Phase 3 clinical trials in combination with other ITP therapies, such as corticosteroids or Promacta (eltrombopag), as well as a standalone treatment.
Regulators in the U.S. and Europe have granted ianalumab orphan drug status for ITP, a designation that aims to support the development of treatments for rare diseases.
In addition to ITP, Novartis is testing the therapy in Sjögren’s disease, systemic lupus erythematosus, and other B-cell-driven autoimmune conditions.
Therapy snapshot
| Treatment name | Ianalumab |
| Administration | Intravenous infusion, potentially in combination with other ITP therapies |
| Clinical testing | Currently in Phase 3 testing |
How will ianalumab be administered in immune thrombocytopenia?
In clinical trials testing ianalumab in people with ITP, participants have received intravenous infusions containing varying doses of the therapy once monthly.
In some studies, participants also received simultaneous treatment with corticosteroids or Promacta. Corticosteroids are often used as a first-line treatment for ITP, whereas Promacta is approved for people with ITP who haven’t responded well to other therapies.
Ianalumab in immune thrombocytopenia clinical trials
Ianalumab is being tested alone and in combination with other therapies in adults with primary ITP across several clinical studies, all of which are part of the VAYHIT clinical trial program.
- VAYHIT1 (NCT05653349), a Phase 3 trial focusing on patients who require first-line treatment with corticosteroids. Participants are receiving one of two doses of ianalumab, or a placebo, once monthly, in addition to corticosteroids. The primary outcome metric is the time period between the beginning of the study and treatment failure. Platelet counts dropping below a reference value of 30 G/L, need for rescue treatment or second-line treatment, or death are considered treatment failure.
- VAYHIT2 (NCT05653219), a Phase 3 trial enrolling patients who have failed to respond to first-line treatment with corticosteroids. In addition to four monthly doses of ianalumab or a placebo, participants were given Promacta. Recently announced preliminary results have shown that the trial met its primary goal, with ianalumab delaying treatment failure, which was defined in a manner similar to that in VAYHIT1. Individuals in the ianalumab groups also experienced a significantly higher rate of sustained improvements in platelet counts at six months.
- VAYHIT3 (NCT05885555), a Phase 2 trial involving patients who had already been treated with at least one corticosteroid and one thrombopoietin receptor agonist (TPO-RA), a category of ITP treatments that includes Promacta. All participants received the same dosage of ianalumab, with a total of four doses administered over four months. If clinically indicated, they might also continue receiving treatment with a corticosteroid and/or TPO-RA, provided they were on a stable dose for at least 14 days before starting treatment with ianalumab. In an interim efficacy analysis of 10 participants, 50% achieved platelet counts above a healthy threshold (50 G/L) at two consecutive assessments without additional treatments. Of these, four participants took a TPO-RA concurrently, and one was on ianalumab alone.
A separate Phase 2 trial, VAY RE-HIT (NCT07039422), will examine whether a second course of treatment with ianalumab can help people who experienced treatment failure in VAYHIT1 or VAYHIT2 after having benefited from the therapy.
Ianalumab side effects
Side effects reported in the VAYHIT3 trial testing ianalumab in ITP patients who had failed to respond to at least two prior lines of treatment included:
- infections
- infusion-related reactions
Bleeding Disorders News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by